Diabetes healthcare is being boosted by greater access to and understanding of what these little bits of wearable wellness can do for users.
When we published a feature, A Sensor Adventure, in the summer of 2017, there really weren’t that many about, they were rarely seen and not widely understood in terms of the benefits they can bring to individuals. Many people have had to self-fund, but at least more people have used sensors and started to see their value in terms of improved diabetes control.
The main contenders 18 months ago were Medtronic’s Enlite sensor, back then mainly used in combination with the company’s 640g insulin pump, and there was Dexcom’s G5 – since replaced by G6 – that was used by non-Medtronic pump users. And there was Abbott’s alternative, the FreeStyle Libre, plus a few people operating IDY (hacked) sensors, still a growing community. Since then, both Medtronic and Dexcom have broadened out with offerings that work for people using multiple daily injections (MDI). Plus, Abbott’s FreeStyle Libre has been accepted nationally as a strip-free alternative to blood testing. There are new features on all of these products, but beyond that in the next few months there should be at least two or three new contenders on our shores, bringing the choice up to seven or eight. This should serve to increase uptake and presumably drive down prices.
In case you’re wondering what the fuss is about CGM, here is a reminder: CGMs have a set of alarms that can be set up, alerting the wearer to trends of blood glucose going up, or down. The idea being that if you can detect and prevent these we can go a long way toward reducing the long-term complications that come from high and low blood sugars.
Back in a flash
CGM is not Flash Glucose Monitoring, which is the technology behind Abbott’s FreeStyle Libre sensor and which requires the user to swipe the sensor in order to pick up blood glucose data. This means there is no transmitter worn on the sensor, keeping it a smaller unit to attach to the body. It’s also a slightly more affordable technology, partly as each sensor lasts 14 days. The version currently available in the UK does not have alarms on it, though that is likely to change sometime this year when a newer version comes out. Full CGM – Dexcom, Medtronic and Medtrum at present – includes a transmitter, with each sensor lasting seven days.
Alarms, which can be both annoying and tiring, are also utterly invaluable when it comes to giving the wearer the ‘heads up’ as to the status of their blood glucose control. An alert can warn if you are trending too high, or – even more importantly – too low, giving you the chance to take action and avert a hypo.
Improvements in as well growing understanding of the benefits, combined with greater competition in the arena, is leading to better access to sensors – see the report on the recent NHS Long-Term Plan, which includes specific reference to sensor provision for people with diabetes who qualify.
Increased use of sensors is an important step on the way to a bigger goal, that of closed loop technology – also referred to as the ‘artificial’ or ‘bionic’ pancreas. Increasingly, CGM data is capable of being beamed directly into a connected pump system, which can be set to automate insulin delivery (closed loop). In the last issue we did an extensive feature on the Medtronic Minimed 670g insulin delivery system, the first self-adjusting closed loop pump. That’s just the start; all pumps in the future are likely to come with a sensor combination, so getting used to wearing one is one way to get further along that path.
Body of proof
Proof of positive impact of CGM for diabetics on injections was shown via the DIaMonD study (Multiple Daily Injections and Continuous Glucose Monitoring in Diabetes), the first-of-its-kind study that demonstrated the impact of CGM on HbA1c and hypoglycemia in participants on a multiple daily injection (MDI) insulin regimen. Published in January 2017 in Journal of the American Medical Association (JAMA), the study showed Dexcom CGM System users on MDI achieved a 1% average reduction in HbA1c after 24 weeks of regular use. In addition to better glucose control, participants also increased time spent in target range and spent less time in hypo and hyperglycemia, compared to those using a meter to monitor their glucose.
Furthermore, news that costs for using Dexcom G6 are to be covered by US health insurers is also proof that CGM works. The system will be covered for Medicare beneficiaries, having met the category requirements for therapeutic CGM systems by the US Centers for Medicare & Medicaid Services (CMS). Coverage for therapeutic CGM includes certain beneficiaries who have either Type 1 or Type 2 diabetes and intensively manage their insulin. The company expects to begin shipping product to Medicare customers in the first part of 2019.
In the loop
It should be noted that closed loop pumps work off sensor data – essentially these will be the pumps of the future. Very newly available in the UK is Medtronic MiniMed 670g closed loop insulin delivery system, which can be set to automatically deliver or suspend insulin based on readings from an Enlite CGM sesnsor.
Also, in February 2019 American company Beta Bionics announced it had gained further funding for its iLet ‘bionic pancreas’ for people with Type 1 diabetes. Using machine learning and data from a CGM device, the algorithms calculate and dose insulin and/or glucagon as needed.
Help with access: If you need to know more about, or need help to put your case forward in getting diabetes technology, contact Input Diabetes who may be able to help.
THE SENSOR SCENE 2019
This overview shows sensors listed in roughly alphabetical by product name, or by company name if the product name is not yet known. Cost indications are indicated where available.
Ascensia
This month Ascensia Diabetes Care announced it had entered into a global alliance with Zhejiang POCTech, Co., Ltd (POCTech), a developer and manufacturer of continuous glucose monitoring (CGM) systems.
The intention is to bring to market a state-of-the-art CGM product that improves on the current GCMs available. As part of this agreement Ascensia will commercialize a version of POCTech’s current CGM kit, distributing the product in the second half of 2019.
Ascensia is responsible for the development of the Contour Next blood glucose monitoring range of meters. The company will be the exclusive distributor for POCTech’s devices in an initial 13 markets, which are yet to be confirmed.
This agreement will leverage expertise from both companies to build and manufacture next-generation products with enhanced capabilities and features that are designed to more effectively meet the needs of people with diabetes.
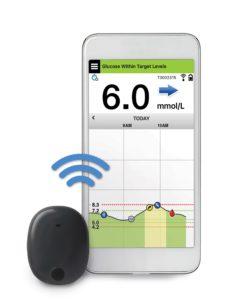
 G6 from Dexcom
G6 from Dexcom
The Dexcom G6 CGM sensor lasts 10 days and no confirmatory fingersticks are required (neither to dose, nor for calibrations).
The G6 works with a smartphone app, so you can see your results on your phone’s screen whenever you wish, and get alerts sent to you for impending highs or lows.
It includes an ‘Urgent Low Soon’ alert, giving advance notice of a potential hypo, allowing time to intervene earlier to avoid going low. This is just one of a range of customisable alerts: users can fully customise their alerts to make them more relevant to their requirements.
Dexcom’s G6 sensors are licensed for anyone with diabetes more than two years old. There are also options to share data from within the device, so parents, partners, carers or healthcare professionals can get access to the alerts too.
A one-month starter kit is available for £159 (x3 sensors and a transmitter), with monthly contracts available from £159.
 EVERSENSE from Accu-Chek
EVERSENSE from Accu-Chek
The Eversense system continually measures interstitial fluid glucose levels in adults with diabetes for the operating life of the sensor, which is 180 days (six months).
Although the CGM is implanted the user wears a reader on the skin, which delivers glucose trend information and includes alarms for the detection and prediction of episodes of hypo and hyper. This data can be transmitted via an app to a smartphone.
The aim is that the system will allow users to manage their diabetes with more flexibility, confidence and discretion. Also, the system needs calibrating twice a day with a blood glucose reading.
The Eversense sensor is inserted in a clinical environment, the implanted sensor is powered wirelessly by the transmitter placed on the arm directly over the sensor. There are only a few centres where implants can be done.
To find out more contact Accu-Chek direct or talk to the people at London Diabetes Centre.
ENLITE from Medtronic
Medtronic’s Enlite sensor works with the company’s Minimed insulin pump series. It can also be used as part of Medtronic’s Guardian Connect alongside other pumps or by people on MDI as a stand-alone system.
The purpose of the new Medtronic Guardian Connect is for people who either don’t want to pump, can’t get a pump, or have Type 2 diabetes and are not being encouraged to use an insulin pump.
Therefore, the Medtronic Guardian Connect does not work with an insulin pump but with a smartphone. This consists of an Enlite sensor worn on the body that uses a Guardian Link to transmit glucose results every five minutes via Bluetooth to a free app on a smartphone.
This means easy visibility of data for the user (or interested parties — the system can also be made to send SMS alerts to parents, partners or HCPs.).
Enlite sensors need calibrating at least twice a day. If you’re using the app, you just plug the calibration from your blood test meter into the app.
Using the Enlite with either the 640g or 670g, or as part of Guardian Connect, comes with a system of alarms that can be adjusted around an individual’s needs. You can also set up your own alerts and you’ll see arrows showing you if your readings are trending up or down. These have been proven to increase time spent in range, helping users to eliminate being too high or too low.
 FREESTYLE LIBRE from Abbott
FREESTYLE LIBRE from Abbott
The aim of Abbott’s FreeStyle Libre system is to eliminate the need for routine finger prick testing. The popularity of this easy-to-use product has seen an impressive uptake, with plenty of customers happy to scan themselves to see what’s happening with their control.
One bonus is that parents are able to go into their sleeping child’s room at night and scan them without waking them or having to prick their fingers to do a test.
Part of the company’s commitment has been to educate both healthcare professionals and users to make the best of this technology, which it does via its Freestyle Libre Academy.
If you are interested in accessing Libre sensors you need to sign up to the waiting list. A starter pack costs £159.95 for one reader (which should last years) and two sensors which each should last 14 days. Subsequent readers or sensors cost £57.95.
The Libre Link app, which allows you to scan the sensor with your smartphone, is free to download and use.
SUGARBEAT from Nemaura
Nemaura has developed predictive algorithms to support alarm functions in its wearable SugarBeat glucose monitoring system. The British company is expecting to win CE Mark approval any day now, at which point it will launch in the UK. A small group of users willhave access to the device initially, this will then be followed up with a full scale launch on a gradual basis across the UK and into other major European territories.
 TOUCHCARE by Medtrum
TOUCHCARE by Medtrum
Medtrum supplies both an insulin patch pump and a CGM sensor for people with diabetes who might need one or the other, or both.
The A6 Touchcare system comes complete with apps as well as a comprehensive website that carries tutorial videos on aspects of using the pump and sensor. Data picked up by the associated smartphone app includes trend graphs to show if blood glucose is rising or falling.
There are x4 sensors per box, each worn for seven days, so one box lasts a month.
The CGM transmitter is rechargeable and the sensors take a measurement every two minutes, the most frequent of any glucose-monitoring sensor currently available.
For those who wish to self-fund there are two options for starter packs. Option 1 is one sensor plus one transmitter for £135. Option 2 is for a box of sensors plus one transmitter for £225. There are no annual commitments for either option, and ongoing costs are £35 for one sensor that will last a week, or £140 for a box.
News items and features like this appear in the Desang Diabetes Magazine, our free-to-receive digital journal (see below). We cover diabetes news, diabetes management equipment (diabetes ‘kit’ such as insulin pumps and continuous glucose monitoring equipment) and news about food suitable for a diabetic diet including a regular Making Carbs Count column. We just need your email address to subscribe you (it’s free, and you can easily unsubscribe should you wish to).